
The New SP Hull LyoStar 4.0 Now Available From Biopharma Group

‘Advancements in Freeze Drying Production & the Impacts on Sale, Sustainability and Compliance’ – Labmate UK & Ireland
Freeze Drying vs Spray Drying – Which is Most Suitable For Your Projects?
The demand for productivity and profitability in the freeze drying, also known as lyophilisation, sector is increasing. This increase is driving practitioners in the industry to consider the benefits of freeze drying compared to spray drying. In this article, we have therefore played a bit of a ‘devil’s advocate’ when considering the advantages of both to help you choose which is best for your project and product.
 Which Should You Choose?
Which Should You Choose?
Freeze drying is widely recognised in the biopharmaceutical industry as the preferred method for preserving a wide range of pharmaceutical formulations and biologicals. This drying method is particularly beneficial when stability in the liquid state is inadequate, storage requirements are too stringent, or a solid form of the product is desired to improve shelf life or enable shipping between differing climates and environments.
Freeze drying is certainly the established ‘go to’ drying process across a variety of materials and applications. However, owing to the costs, availability of APIs and sometimes processing and production volumes, evaluation of alternative methods is conducted to ensure that the most suitable drying method for the product and/or project is used. The most popular alternative to freeze drying is spray drying.
Spray drying also offers a variety of benefits. Despite being a comparative newcomer in the industry versus freeze drying, it showcases capabilities to work with higher throughput amounts at a scalable level (continuously rather than batch by batch). As a result, spray drying can also be seen as a viable option for product drying, subject to the contextual processing requirement and product application.
The Drying Process
The principle of freeze drying involves the initial freezing of a product, in a controlled manner, to manipulate the ice crystal structure. It is then placed in a vacuum where sublimation (or primary drying) takes place to remove the unbound water.
Next comes secondary drying to sublimate the bound water, taking the material down to a user defined residual moisture level. The target at this stage is bound rather than unbound/free water, therefore more energy is required to drive the process by raising shelf temperatures to +20°C or above. When coupled with a low atmospheric pressure, it causes the ice to turn directly into water vapour (by passing the liquid phase); this process is called sublimation.
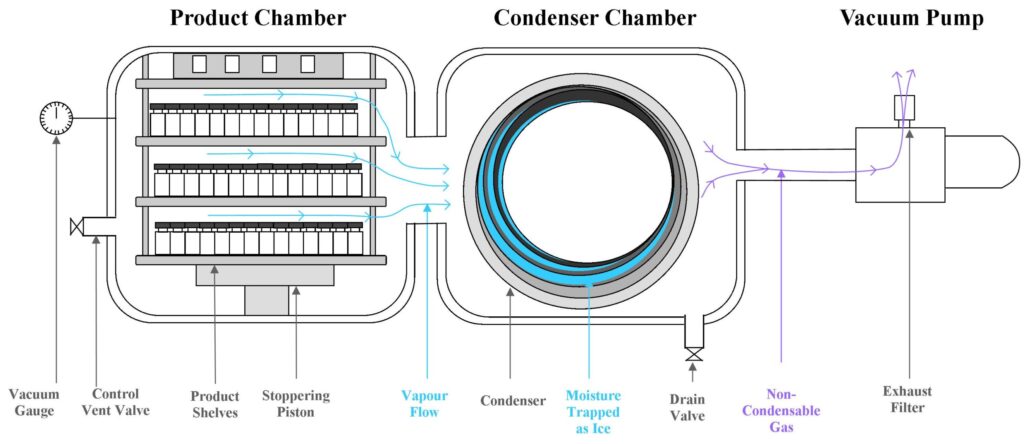 Subject to the nature of the sample being dried in a given lyophilisation cycle, the delicate balance between temperature and vacuum is vital to ensure that a successful batch, with no impact on product efficacy and viability, is produced post drying. To achieve this, differing type of products and the volumes thereof, may need to undergo freeze drying conditions ranging from 12 hours to 5 days, based on their inherent sample properties.
Subject to the nature of the sample being dried in a given lyophilisation cycle, the delicate balance between temperature and vacuum is vital to ensure that a successful batch, with no impact on product efficacy and viability, is produced post drying. To achieve this, differing type of products and the volumes thereof, may need to undergo freeze drying conditions ranging from 12 hours to 5 days, based on their inherent sample properties.
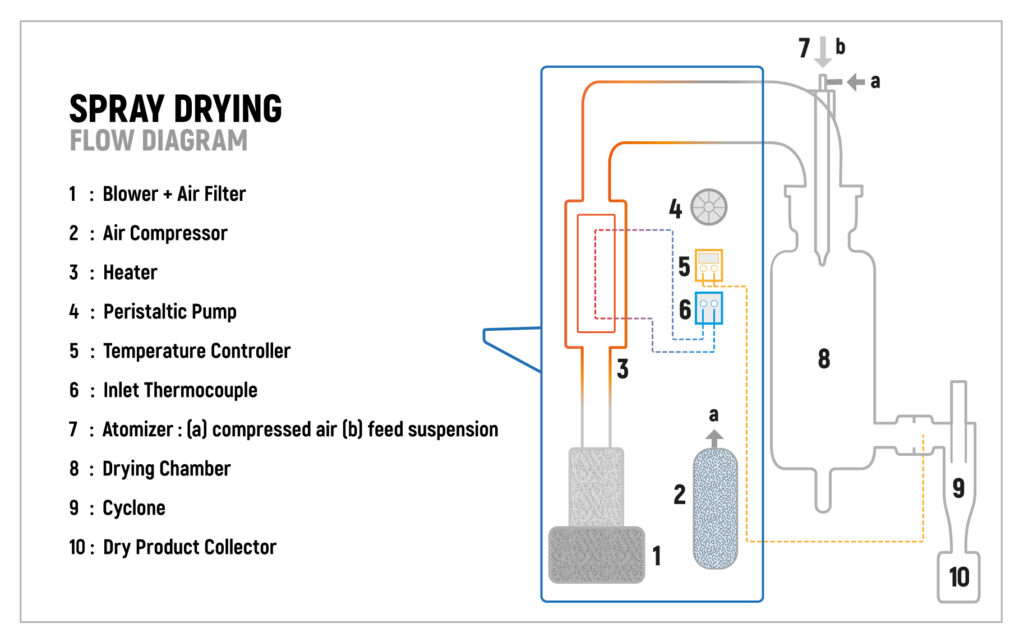
Spray drying is generally considered to be a simpler (and faster) process, involving the conversion of a liquid formulation into a dry powder in a single step. The solution is atomised into fine droplets which are quickly dried straight after in a large chamber using warm gas. The resulting dry particles are then collected with a cyclone.
Although faster, and less expensive than freeze drying as a rule of thumb, one of the significant drawbacks are the high processing temperatures and shear forces it demands. These, of course, are what many customers seek to avoid in the stringently controlled pharma and biotech industries where viability and process parameters for efficacy relate to sensitive APIs and formulas; spray drying therefore, is more suitable for use with reasonably hardy products.
Product temperatures in freeze drying are generally below 0°C in primary drying and 20-30°C during secondary drying, whereas product temperatures in spray drying are regularly above 80°C. The direct impact of working with these higher (80°C) temperatures, can be an overall loss of sample quality post drying in terms of inherent product characteristics such as:
- Efficacy/viability
- Taste/smell/colour
- Consistency
- Nutritional value i.e. nutrients in food related products
- Biological yield – greater level of log reduction of cells i.e. bacterial
- Degradation of proteins



Use in the Industry
Both processes can be used for a vast range of applications. For example, freeze drying is typically sought out for the preservation of different cell types, fine chemicals, laboratory reagents and injectable vaccines, as well as for the food industry & dairy products. Because it is typically performed with product directly filled in vials or other containers, this processing method is best suited for formulations that do not require further processing after drying; additionally, vials can be sealed in-situ of the freeze dryer, thus avoiding potential contamination when the cycle is complete.
Spray drying, on the other hand, is more commonly associated with bulk processing rather than vial based processing. However, it is a common misconception that spray drying is only a suitable process for food and robust bulk pharmaceuticals, where contemporary research suggests it may be a valid methodology for use with some complex products i.e. microencapsulated bacteria and nano particulates, for example.
 Efficiency, Quality and Cost
Efficiency, Quality and Cost
It is widely accepted the costs associated with spray drying are less than those of freeze drying, which has made the technique one of interest for certain markets. More open to larger throughput potentials, spray drying can be deemed a ‘continuous process’, unlike the batch format associated with freeze drying.
Moreover, it is the robust quality of product where freeze drying’s strength lies. Accurate control of low processing temperatures minimises any risk of intrinsic products properties, such as collapse, eutectic melt, or glass transition temperatures being exceeded, making freeze dried products processed to the highest quality. The shear stress biopharmaceuticals can be exposed to during spray drying, combined with the required high processing temperatures can destabilise compounds such as proteins and damage product properties, ultimately diminishing product efficacy.
In conclusion, both methods of product drying process can be effective when used correctly, and for the appropriate products. In order to achieve optimal results in product processing that includes a drying stage, ultimately the deciding factor for which method will be best for a project, or ongoing processing need, will be the quality of the end product and how it will reach the end users.
Biopharma Group has been an industry leading expert in drying technology field for over 30 years. Our dedicated lyo lab with dedicated scientists, based in the UK, houses both freeze drying and spray drying instrumentation at R&D scale. This allows us to offer the best drying process solution to suit your needs and to perform unbiased comparisons between both techniques, for your reference to support the development of your product now and in the future.
