Laboratory/ Benchtop Scale Freeze Dryers
AdVantage Pro
Benefits offered by the Advantage Pro
- Designed with both scale-up and scale-down, in mind,
- Cascade refrigeration provides a -82°C large 6L capacity condenser coil, enabling lyophilisation of a wide variety of products and solvents.
- Optimal heat transfer - silicone flooded shelves mean the AdVantage Pro maintains a precise level of temperature control (+/- 1.0 °C).
- Fully customisable - up to ten thermal treatment and twelve drying steps for the ultimate recipe customisation.
-
Secure data storage and trending software option - ethernet-enabled for effortless data sharing. Developed to meet the lyophilisation requirements of today’s pharmaceutical and biotechnology applications.
- Compact design and small footprint - Only 56.5 cm wide, yet with up to 2,766 cm2 of usable shelf area, the AdVantage Pro enables product to be safely processed using minimal bench space.
AdVantage Pro Freeze Dryer Trending Software
Supplied pre-loaded on a dedicated laptop PC offering plug and play ethernet connectivity to your AdVantage Pro freeze drying unit (when purchased with an AdVantage bench top unit – retrofit options are available). This iFIX-based software package provides graphics and interactions complimenting those found in the Merlin and LyoS series of Pilot scale control and trending software so simplifying upscaling from bench top processes.
What it has:-
- Recipe manager with 16 recipes
- Continuous historical data trending
- Proficy Historian data archiving
- Data export capability
Click Here For Software Options


New to Freeze Drying?
Click on the button below for our Product Selector
Pilot & R&D Freeze Dryers

Genesis & Ultra
Ultra Prime 50 from SP - An ATS Company
The Ultra Prime 50 is a state-of-the-art freeze dryer with customisable options including up to 15 shelves, for R&D through to small-batch production and designed for superior performance, repeatability, and sustainability. Incorporating advanced LyoLogic HMI technology, the Ultra Prime 50 offers enhanced cycle efficiencies, an innovative baffle system for improved ice capture on the condenser, and a space-efficient footprint. These advancements reduce energy consumption, operational costs, and environmental impact.
Key Features:
- Innovative LyoLogic HMI – A new controls platform with increased functionality.
The LyoLogic control system is designed to meet the lyophilization requirements of modern pharmaceutical and biotechnology applications. Its user-friendly interface provides intuitive controls that simplify the freeze-drying process while ensuring precision and flexibility. - Upgraded GWP (Global Warming Potential) Sustainability
Ultra Prime uses refrigerant gases with GWPs of just 2 and 6, a significant improvement over previous systems, which had GWPs of 1,774 and 13,396. This reduces the environmental footprint. - Superior Performance
A single product chamber design allows for larger batches and greater product uniformity. - Space-Saving Design
A compact, free-standing, mobile design enables easy installation and optimal use of lab space. - Efficient Smooth-Walled Condenser
The smooth-walled condenser minimizes contamination risk and ensures efficient freeze-drying cycles for optimal results.
The Ultra Prime 50's compact design still packs a punch with shelf temperatures as low as -70ºC and condenser temperatures down to -85ºC, supporting a wide range of applications. With standard options and 50L condenser capacities, you can rest assured that there’s an Ultra Prime configuration to meet your requirements, whether that’s high moisture throughput capacity for high moisture products such as vials or bulk liquid API, or a high shelf surface to condenser surface ratio, for low moisture applications such as diagnostic kit development and tissue banking.
Available with a Merlin or LyoLogic control system.


Benefits offered by the Genesis & Ultra
- A perfect solution for operators seeking versatile freeze dryers capable of cross-over between applications / batches us at the pilot and small scale production, level
- Flexibility - an extensive choice of powerful options and add-ons enables easy scale-up from research to full scale production.
- With over 100 flexible configurations - the ideal choice whether you’re in academic research, a biotechnology or pharmaceutical company, part of a government agency, or working in industrial manufacturing.
-
Easy to customise -up to 6 shelves (Genesis) or 15 shelves (Ultra) for personalised configurations. Ultimate versatility with bulk or stoppering configurations, air cooled or water cooled, can accommodate both ‘wet’ and ‘dry’ vacuum pumps to meet the preference of the user
- Range of options - from manual controls to the latest automated control with cGMP data monitoring and automation applications for 21 CFR Part 11 requirements
- All controls and features are match those used in performance matched SP VirTis brand industrial lyophilisers, with shelves and chambers engineered using premium chemical resistant 316L stainless steel. Thereby providing advanced, reliable and scalable lyophilisation.
Click Here For Software Options


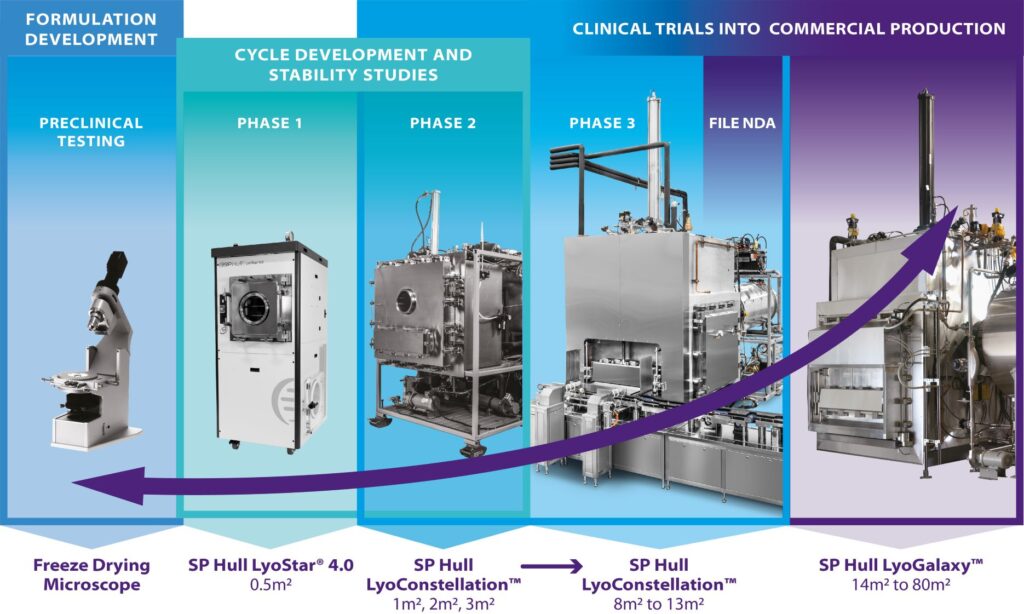
LyoStar 4.0
Benefits offered by the Lyostar 4.0
- Data rich environment: Ensuring product processing success, even if deviation from 'design space' occurs
- Scalability: LyoStar 4.0 offers the ability to scale up from R&D to Production scale and back again
- Faster and more efficient: SMART Technology & Controlyo reduce processing and reconstitution times significantly
- Reduced contamination risk: The non-invasive sensors of LyoFlux TDLAS and wireless probes of Tempris offer a reduced risk of cross-contamination
- Eco-friendly: LyoStar 4.0 uses a 'greener' refrigerant gas that has been selected due to its lower global warming potential
- 21 CFR Part 11 Compatible: The flexibility to scale-up or down is an important aspect in the process for developing pharmaceutical compounds. The LyoStar 4.0 has the same 21 CFR Part 11 compatible software as the LyoConstellation, making scale-up, or down, an easy transition.
Ideal for rapid lyo cycle development, optimization and scale-up, the LyoStar 4.0 incorporates the latest innovations in freeze drying technology to ensure precise process control and reliability. Enabling this are a number of upgradeable PAT tools including: LyoFlux® TDLAS sensors for vapor mass flow measurement, AutoMTM/SMART™ technology – a primary drying cycle optimization tool, ControLyo® Nucleation Technology for accurate freezing point control, a 3D modelling package for computational fluid dynamics and process monitoring, plus Tempris® wireless sensors for real-time product temperature measurement.
Get in touch today
Tel: +44 (0) 1962 841092 | bps@biopharma.co.uk
Production Freeze Dryers
Aseptic Production Freeze Dryers:
In the pharmaceutical and biotechnology industry, devices must be of very high quality with high precision control tools. Biopharma Group has worked for over 30 years with manufacturers recognized worldwide for the quality and technological advances of their industrial freeze dryers. We supply their equipment to the largest industrial groups in the world in the health, life sciences and pharmaceutical sectors
LyoConstellation + LyoGalaxy series of aseptic freeze dryers have been designed with throughput capacity (including automated loading / unloading), product efficacy, cleaning validation, and data integrity 21 CFR Part 11 needs, in mind. Our host of PAT technology offers an abundance of real time data to support the day-to-day operations as well as, the QP for those occasions when deviations may arise.
You may also be interested in:
Automation Options:
Aseptic Processing Lines Fill-Finish Solutions
White Papers:
Technologies & PAT Tools to Overcome Lyo Challenges
Putting Fill-Finish Customization into your Line of Sight
Also:
Link to SP Hull Freeze Dryer Loading & Unloading Systems by ATS Life Sciences Scientific Products
LyoConstellation Spec Sheets:
Production Freeze Dryers (non-aseptic):
The Benchmark production freeze dryer range comprise custom built solutions to meet your specific process needs.
- Optimised shelf designs – varied shelf sizes and inter-distances provide maximum processing capacity within a compact / versatile footprint, with shelf area up to 42m²
- Shelf temperatures to -70’C and condenser temperatures to -75’C, making the Benchmark an ideal choice of equipment no matter your process formulation
- Internal or external condenser coils, to fulfil your exact requirements
- Clean-in-Place, CIP, is also available, to support the cleaning activity of your system, if required
- Data integrity – software offering full 21 CFR Part 11 compliance
- Machine redundancy – option to select hot swappable and redundant refrigeration components helping to minimise any downtime, offering uninterrupted use and easy component management
- Cleanroom "through-the-wall" design compatible
- Our team of in-country engineers offer companies and users alike the option of machine validation such as SAT / IQOQ, and on-going system servicing
Benefits offered by our non-aseptic production freeze dryers
- Production Freeze Dryers designed for a variety of life sciences applications:
- Diagnostic Kits & Bulk Reagents
- Bulk Collagen & Tissue Products
- Nutraceuticals
- API (Active Pharmaceutical Ingredients)





Freeze Drying Expertise
It is our extensive freeze drying and project management experience that allows us to select the system that is best suited to your specific production process needs. We have installed devices with specifications and configurations as varied as they are numerous: CIP and SIP systems for cleaning and sterilization, loading systems for rows of products, flap doors (‘pizza oven’ type), automatic loading and unloading (ALUS), all compliant with 21CFR11 international standards and GMP (cGMP and GAMP5). Complete documentation is available for each device, to meet the requirements of users and applicable authorities. We are at your disposal to advise and assist you through your purchasing project and beyond.
Installation and Qualification
Our Technical Service team can assist you with commissioning, operator training and installation qualification to ensure that your system remains in compliance with your practical requirements as well as applicable regulatory standards. We also offer an after-sales service for the regular maintenance for the life-time of your machines.
CDMO Specialists for Lyophilised & Liquid Formulations
Biopharma Group offers scalable CDMO solutions across an array of products & industries. Dedicated in-house scientists provide dried product & liquid formulation analyses including:
- Pre- & post-lyophilisation R&D
- Comparative process studies
- Formulation/cycle development
- Specialist post-process analysis
- Class 7 facility for cyto & high potency product processing
- Non-GMP production & packing facilities
- GMP processing ideal for small batch, sterile, first-in-human trials production
To learn more about our CDMO services, visit www.biopharmgroupacdmo.com
Get in touch today
Tel: +44 (0) 1962 841092 | bps@biopharma.co.uk